|
FluoroNanogold™ probes containing both a fluorescent label and the Nanogold® cluster covalently linked to a targeting molecule have been successfully used for correlative fluorescence and electron microscopy (EM) [1]. However, Nanogold is small (1.4 nm) and requires silver or gold enhancement for visualization [2]. This produces much greater size variability than is found in larger colloidal gold preparations. Silver enhancement may lead to non-specific background; and silver can be chemically etched by osmium tetroxide (OsO4) fixation leading to signal loss [2]. We have prepared covalently linked combined fluorescent and larger gold nanoparticle probes for correlative microscopy that can be directly visualized by EM without the need for autometallographic enhancement. Gold nanoparticles, 2 nm to 40 nm in diameter, were stabilized and functionalized using a self-assembling coating comprising a hydrophobic chelating thiol domain to seal the gold surface from aqueous media, and hydrophilic terminal functional groups (polyethylene glycol, PEG) that biocompatibilize the gold nanoparticles and enable site-specific covalent probe conjugation. This provides high stability and a wide choice of surface properties and conjugation reactions.
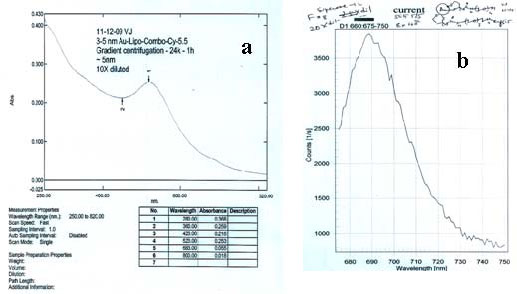
5 nm gold nanoparticles were prepared by direct reduction of aqueous solution of gold (III) salt with sodium borohydride in the presence of a mixture of solubilizing non-functional and protected aminefunctionalized thiols, followed by purification using sucrose density gradient (10-30%) ultracentrifugation (Ti-41 rotor, 26,000 rpm, 45 min, 5 ºC; the intense red band that travels ~ 4 cm from the top contains 5 nm gold). Amine-functionalized gold particles were prepared by deprotection in methanolic hydrochloric acid. Specific reactivity was demonstrated by reaction with commercial N-hydroxy succinimido-(NHS) Cy-5.5 fluorescent dye. This conjugate was further purified by Superose-12 size exclusion chromatography (Figure 1). Unstabilized particles of this size are precipitated by 0.1 M sodium chloride, but the coated particles remained dispersed and suspended, even in 1.0 M sodium chloride solution. They were repeatedly centrifuged and completely resuspended without leaving any solid precipitate behind; identical treatment of conventional protein or macromolecule-stabilized colloidal gold conjugates always results in loss of a fraction as an insoluble pellet that cannot be resuspended.
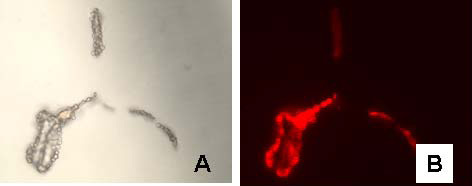
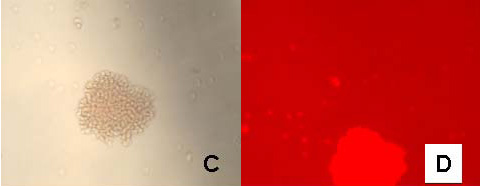
Combined 5 nm gold-secondary antibody-Alexa Fluor 594 conjugates were prepared by covalently linking NHS- and Maleimido- activated 5 nm gold particles to the secondary antibody molecule, IgG and F(ab’) respectively. NHS-modified Alexa Fluor 594 dye was then reacted with the conjugate. The product was purified using sucrose density gradient ultracentrifugation followed by gel filtration as above. The anti-rabbit-F(ab’) conjugate was used as a secondary probe against a polyclonal rabbit anti-red blood cell antibody to label sheep red blood cells in suspension and the labeling was observed by fluorescence microscopy using Nikon G-2A filter set (Figure 2). The anti-mouse-IgG conjugate was used as a secondary probe with mouse anti-human AE1/AE3 primary that produced clear staining of cytokeratin in human tonsil tissue (Figure 3). The relative quantum yields for the fluorescent gold conjugates were 22-35% of the corresponding commercial dye labeled secondary antibodies.
Figures
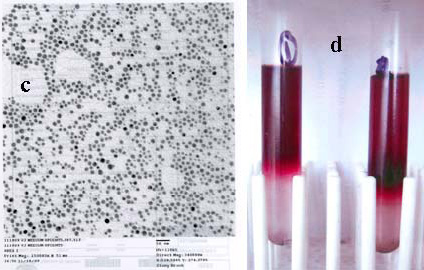 |
| Figure 1: UV-visible, fluorescence (Ex 660 nm) spectra, and TEM (a, b, and c) of fluorescent 5 nm gold-Cy-5.5 conjugate purified by density gradient ultra-centrifugation (d) followed by size exclusion chromatography (vide supra). |
 |
| Figure 2: Bright field (A) and fluorescence images (B) of sheep red blood cells labeled with 5 nm gold anti-rabbit-F(ab')-Alexa Fluor 594; C and D are images for control cells in which the primary rabbit anti-sheep red blood cell antibody was excluded. |
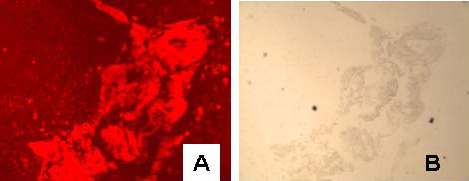
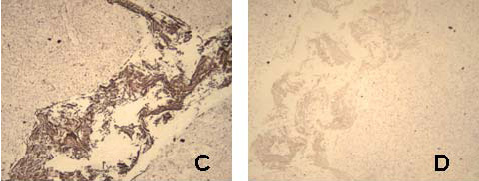 |
| Figure 3: Fluorescence (A), bright field (B), and bright field image following silver development (C) of cytokeratin stained tonsil tissue with mouse anti-human AE1/AE3 primary and 5 nm gold anti-mouse-IgG-Alexa Fluor 594 secondary antibody; D is corresponding control slide in which the primary mouse anti-human antibody was excluded. |
References
- (a) F. Braet et al, Microsco. Res. Tech. 70 (2007) 230; (b) J. M. Robinson et al, J. Histochem. Cytochem. 49 (2001) 803; (c) R. D. Powell et al, Microsc. Res. Tech. 42 (1998) 2.
- J. F. Hainfeld and R. D. Powell, Cell Vision, 4 (1997) 408.
- This research was supported by NIH SBIR grant 1R43 EB008621.
|