The small size of our 1.4 nm Nanogold® gold nanoparticle probes, their lack of aggregation and their consequent ability to access nuclear antigens makes them an excellent reagent for ultrasensitive in situ hybridization detection of specific DNA sequences both with and without in situ PCR.
Contents
Advantages
- More sensitive than NBT-BCIP alkaline phosphatase or peroxidase in combination with label amplification single gene copy resolution.
- No need for the more lengthy PCR procedures for most cases.
- Avoid false positives found with PCR due to mispriming and amplicon diffusion.
- Black color easily seen with standard bright field microscope; no expensive fluorescent optics required, or problems with autofluorescence and loss of signal from bleaching.
- Black color compatible with full strength standard cell and nuclear stains (H & E, nuclear fast red, methyl green) which greatly improves morphological assessment.
- May be used for EM studies as well.
Complete protocols for In Situ hybridization with Nanogold®-Streptavidin, both with and without the CARD system, are given separately. In addition, the current protocols used by the G. W. Hacker group are now available online.
© Eaton Publishing Co.; used by permission. |
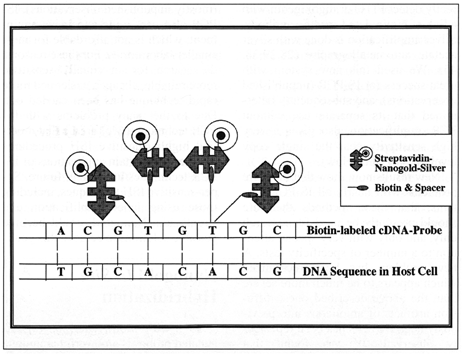
Diagram 1: Schematic representation of the Nanogold-Silver In Situ Hybridization technique. The DNA sequence in the host cell is detected by hybridization with a biotin-labeled cDNA probe. Biotin bound covalently with spacer arms is then detected using streptavidin-Nanogold and subsequently silver-enhanced using silver acetate or silver lactate autometallography. For simplicity, only four such biotin and streptavidin-Nanogold molecules are shown.
Hacker, G. W., Hauser-Kronberger, C., Zehbe, I., Su, H., Schiechl, A., Dietze, O., and Tubbs, R.; Cell Vision, 4, 54-65 (1997).
|
| Detection of the Human Papillomavirus type 6/11 genome using this method is shown below; as can be seen, the Nanogold®-silver staining gives a dense black signal which in many cases is clearer than that obtained using conventional enzyme substrates. |
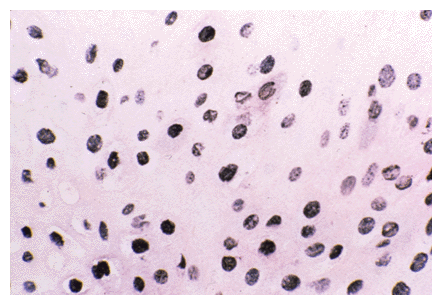 |
HPV 6/11 in condyloma acuminatum detected by Nanogold-silver ISH. In this case, strong labeling was obtained. Most infected nuclei showed dense black or granulated black staining. A few nuclei also showed single black spots of labeling, indicating the presence of very low copy numbers of this DNA virus. Staining was was as described in the protocol; formalin-fixed, 7-micrometer-thick paraffin section, counterstained with hematoxylin and eosin. Magnification, X 285.
|
CARD - CAtalyzed Reporter Deposition (also known as TSA®, or Tyramide Signal Amplification; kits available from NEN Life Sciences) - is a system where a streptavidin-linked peroxidase molecule is used to catalytically accummulate biotinylated tyramide at a site of interest. This inserts an additional amplification step into the procedure before detection with Nanogold®-streptavidin and silver enhancement; the result is a massive increase in sensitivity, which can result in the detection of single-copy genes without the difficulties associated with PCR.
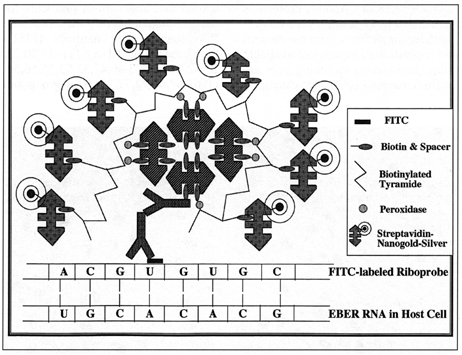
Diagram 2: Schematic representation of the CARD-Nanogold-Silver In Situ Hybridization technique. The RNA sequence in the host cell is detected by hybridization with a FITC-labeled riboprobes. A first antibody layer directed against FITC then binds to the FITC molecule used as the nucleic acid reporter molecule. The next layer is a secondary antibody against the IgG of the species prviding the primary antibody, labeled with biotin. For simplicity, only one of each of these two antibodies is shown. Biotin is then bound to Streptavidin-biotin-peroxidase (S-ABC-Peroxidase) complexes. Four such molecules are shown, each of them carrying peroxidase. These peroxidase molecules are then catalyzing the accummulation of biotinylated tyramide (catalyzed reporter deposition), which finally leads to a "multiplication" of the biotin label. As a final layer, streptavidin-Nanogold with subsequent silver acetate or silver lactate amplification (autometallography) is used. Each of these final streptavidin molecules can detect one of many molecules of biotin. In this way, an enormous amplification is reached, in many cases leading to single gene copy sensitivity without the necessity of PCR or 3SR techniques.
Hacker, G. W., Hauser-Kronberger, C., Zehbe, I., Su, H., Schiechl, A., Dietze, O., and Tubbs, R.; Cell Vision, 4, 54-65 (1997).
© Eaton Publishing Co.; used by permission.
This procedure was used for the detection of HPV-16, a cDNA virus, in SiHa cells cultured from cevical cancer specimens. These cells are known to contain only one to a few copies of the viral DNA; Nanogold®-streptavidin in situ hybridization in conjunction with CARD gave dense black granular staining of the cell nuclei; the black signal was more readily distinguished than the signal produced by peroxidase-streptavidin. The procedure may be completed in 5 to 7 hours;
Single copies of HPV 16 in SiHa cells detected by CARD-amplified Nanogold-silver ISH. This cell line derived from cervical carcinoma contains only one to a few copies of HPV 16, which appear as single spots by the technique applied (see protocol for details). Cytospin preparation, counterstained with Nuclear Fast Red. Similar staining is obtained in tissue sections of formalin-fixed paraffin material (Zehbe, et al, Am. J. Pathol. 150, 1553-1561 (1997)). Original magnification, X 560.
top of page
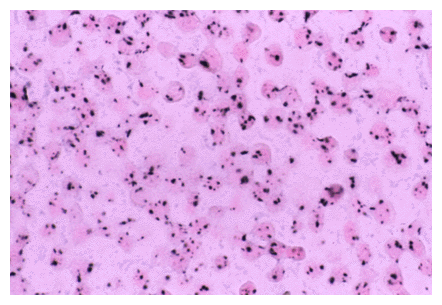
RNA is detected with the same high sensitivity, as shown below:
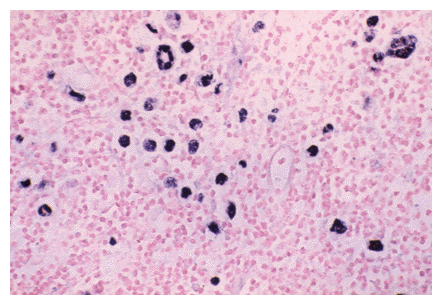
Epstein-Barr virus in Hodgkins Disease; an example of highly sensitive detection using the RNA detection protocol. Original magnification, X 400.
top of page
Nanogold®-Streptavidin also works well in immunolabeling experiments. An example is shown below in which it is used as a tertiary probe against a biotinylated polyclonal secondary antibody against a monoclonal primary to probe for a breast cabcer antigen.
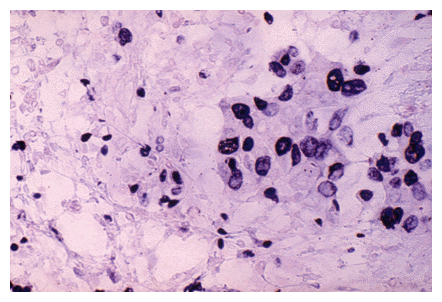
Ki-67 breast carcinoma antigen detected using indirect Nanogold® labeling. Ki-67 antigen was detected using a primary monoclonal antibody followed by a biotinylated goat anti-mouse secondary; this was visualized using Nanogold®-streptavidin (250-fold dilution) followed by autometallography. Original magnification, X 400.
top of page
- Zehbe, I., Hacker, G.W., Su, H., Hauser-Kronberger, C., Hainfeld, J.F., and
Tubbs, R.: Sensitive in situ hybridization with catalyzed reporter deposition, streptavidin-Nanogold, and silver acetate autometallography. Detection of single-copy human papillomavirus. Am. J. Pathol. 150,
1553-1561 (1997).
- Totos, G.; Tbakhi, A.; Hauser-Kronberger, C., and Tubbs, R. R.: Catalyzed Reporter Deposition: A new era in molecular and immunomorphology - Nanogold-silver staining and colorimetric detection and protocols. Cell Vision, 4, 433-442 (1997).
- Hacker, G. W., Hauser-Kronberger, C., Zehbe, I., Su, H., Schiechl, A., Dietze, O., and Tubbs, R.: In Situ localization of DNA and RNA sequences: Super-sensitive In Situ hybridization using Streptavidin-Nanogold®-Silver Staining: Minireview, Protocols and Possible Applications. Cell Vision, 4, 54-65 (1997).
- Hacker, G. W.; Zehbe, I.; Hainfeld, J.; Sällström, J.; Hauser-Kronberger, C.; Graf, A.-H.; Su, H.; Dietze, O., and Bagasra, O; High-Performance Nanogold® In Situ Hybridization and In Situ PCR. Cell Vision, 3, 209 (1996).
- Hacker, G. W., Zehbe, I., Hainfeld, J., Graf, A.-H., Hauser-Kronberger, C., Schiechl, A., Su, H., and Dietze, O.: High performance Nanogold®-in situ hybridization and its use in the detection of hybridized and PCR-amplified microscopical preparations; In Proc 54th Ann. Mtg. Micros. Soc. Amer., G. W. Bailey, J. M. Corbett, R. V. W. Dimlich, J. R. Michael and N. J., Zaluzec (Eds.). San Francisco Press, San Francisco, CA, pp. 896-897 (1996).
- Schöfer, C.; Weipoltshammer, K.; Hauser-Kronberger, C., and Wachtler, F.: High-resolution detection of nucleic acids at the electron microscope level - Review of in situ hybridization technology, the use of gold, and Catalyzed Reporter Deposition (CARD). Cell Vision, 4, 443-454 (1997).
- Tbakhi, A.; Totos, G.; Hauser-Kronberger, C.; Pettay, J.; Baunoch, D.; Hacker, G. W., and Tubbs, R. R.: Fixation conditions for DNA and RNA in situ hybridization: a reassessment of molecular morphology dogma. Am. J. Pathol., 152, 35-41 (1998).
 2016 Nanogold®-Streptavidin (NS) 2016 Nanogold®-Streptavidin (NS)
1.4 nm gold particle is covalently attached to streptavidin.
Instructions for use of Nanogold®-Streptavidin
|