Other applications
The following paper appeared in Proceedings of the fifty-seventh Annual Meeting, Microscopy Society of America; G. W. Bailey, W. G. Jerome, S. McKernan, J. F. Mansfield, and R. L. Price (Eds.); Springer-Verlag, New York, NY, 1999, pp. 478-479. Reproduced by kind permission of Springer-Verlag New York.
Combined Cy3 / Nanogold Conjugates for Immunocytochemistry and In Situ Hybridization
Richard D. Powell, Vishwas N. Joshi, Carol M. R. Halsey, James F. Hainfeld,* Gerhard W. Hacker,** Cornelia Hauser-Kronberger,*** Wolfgang H. Muss,*** and Peter M. Takvorian~
Nanoprobes, Incorporated, Stony Brook, NY 11790.
* Biology Department, Brookhaven National Laboratory, Upton, NY 11973
** Medical Research Coordination Center, University of Salzburg, Austria.
*** Institute of Pathological Anatomy, Salzburg Federal Hospital, Salzburg, Austria.
~ Biological Sciences Department, Rutgers University, Newark, NJ 07102.
Fluorescein and the 1.4 nm Nanogold® gold cluster label may be incorporated into a single Fab' immunoprobe by separate cross-linking reactions, to give a probe which labels antigenic sites in a single step for correlative fluorescence and electron microscope visualization.1,2 These probes show high labeling density, labeling a pre-mRNA splicing factor in the HeLa cell nucleus;3 Microtubules were also densely labeled using fluorescence, other optical modalities, and electron microscopy; in a parallel experiment, a 5 nm colloidal gold probe gave only occasional labeling.4 We now describe Fab' and streptavidin probes containing both Nanogold® and the fluorescent cyanine dye, Cy3.
F(ab')2 Goat anti-Mouse IgG and F(ab')2 goat anti-rabbit IgG fragments were reductively cleaved to Fab' fragments using dithiothreitol (DTT) or mercaptoethylamine hydrochloride (MEA), which selectively reduce the F(ab')2 hinge disulfide bonds, with 5 mm EDTA to prevent reoxidation. Fab' fragments were isolated by gel filtration (coarse gel: GH25, Amicon) then labeled with Mono-maleimido-Nanogold® which reacts site-specifically with thiols. Streptavidin was labeled using Mono-Sulfo-NHS-Nanogold® at pH 7.5. Nanogold® conjugates were isolated by gel filtration (Superose-12 column, Pharmacia), then reacted with excess Cy3 monofunctional NHS ester (labeling kit, Amersham Life Sciences) at pH 7.5; dual-labeled conjugates were isolated by gel filtration (Superose-12).
The Fab' goat anti-rabbit conjugate was used as a secondary probe to label a 55 kDa polar tube protein in spores of the microsporida E. hellum, a protozoan parasite which is opportunistic in AIDS patients. The polar tube is a unique apparatus through which infective sporoplasm is transferred to host cells; its identification is diagnostic for these organisms. Fixed E. hellum-Infected RK-13 rabbit kidney cells were incubated for two hours with a polyclonal rabbit anti-E. hellum EH55ptp antibody, followed by Cy3/Nanogold®-labeled Fab' anti-rabbit IgG. After fluorescence microscopy, silver enhancement (LI Silver, Nanoprobes) was performed. The cells were then embedded in Epon 812, sectioned, mounted on grids and stained with uranyl acetate and lead citrate for electron microscopy. The polar tube was shown clearly by fluorescence, and Nanogold® labeling confirmed by electron microscopy (Figure 1).
The combined Cy3 and Nanogold-labeled streptavidin was used for the in situ detection of HPV-16 viral DNA in different cells specimens using the same methods previously described for Nanogold®-silver staining (NGSS) both with and without signal amplification by Catalyzed Reporter Deposition (CARD, also known as Tyramide Signal Amplification, or TSA).5 Some examples are shown in Figure 2. HPV-16 in SiHa cells, which contain only 1-2 copies of the viral DNA, produced clear fluorescence signals when CARD was used in conjuction with the Cy3/Nanogold®-streptavidin; when the biotinylated tyramide reagent was omitted no fluorescence was observed. Silver and gold autometallography yielded intense punctate staining in the light microscope (which was also absent if the biotinylated tyramide was omitted). Similar results were obtained using CaSki cells, which contain more copies of the viral DNA, and signal localization was confirmed by electron microscopy.
- Powell, R. D., et al., Micros. Res. Technique, 42 (1998) 2.
- Takizawa, T., et al., J. Histochem. Cytochem., 46 (1998) 1097.
- Powell, R. D., J. Histochem. Cytochem., 45 (1997) 947.
- Robinson, J. O., et al., J. Histochem. Cytochem., 45 (1997) 631.
- Hauser-Kronberger, C., Cell Vision, 5 (1998) 83.
- This work was partly supported by NIH SBIR grant 4R44 GM56090.
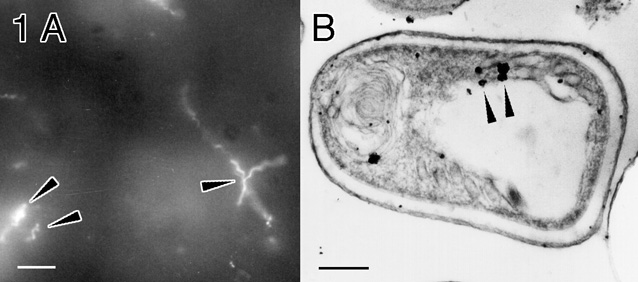
FIGURE 1: E. hellum spores labeled with anti-EH55ptp primary antibody, and Cy3/ Nanogold® Fab' goat anti-rabbit secondary; the coiled polar tube is indicated by arrows.
A: Fluorescence (bar = 10 micrometers).
B: Electron micrograph, after silver enhancement (bar = 0.5 micrometer).

FIGURE 2: In situ hybridization detection of HPV-16 DNA with CARD-Cy3/Nanogold-Streptavidin®. Top row: CaSki cells. (A) fluorescence (bar = 20 micrometers); (B) light micrograph after gold auto-metallography (bar = 10 micrometers); (C) Electron micrograph of same specimen (bar = 1 micrometer). Bottom row: SiHa cells by (D) fluorescence; (E) light microscopy after gold autometallography (bar = 10 micrometers). (F) fluorescence and (G) light micrographs of control with biotin-tyramide omitted (bar = 20 micrometers).
© 1999 Springer-Verlag New York.
Other Applications
|
Product Applications
Research Applications
|
|