|
|
Labeling with Gold Nanoparticles:
Advanced Labeling Calculation Method
for proteins with significant absorption in the visible range
Once you have isolated your gold nanoparticle conjugate, the extent of labeling (the number of gold nanoparticles per biomolecule) can be calculated using the UV/visible spectra of the nanoparticle and the conjugate.
In this section:
Advanced Labeling Calculation Method
Assumptions in calculations:
- We assume that conjugation does not change the UV/visible absorbance spectrum of either the gold nanoparticle or the protein; and
- We assume that the absorbtion of the gold nanoparticle and the protein are strictly additive: the absorbance at all wavelengths for each component is identical to what it would be if the other were not present. For example, even though the gold nanoparticles absorb strongly at 280 nm, the absorbsnce of the protein at 280 nm is unchanged from the value observed for a solution of the same protein without the gold.
The extinction coefficients for the undecagold and Nanogold® nanoparticles have been determined at 280 nm and 420 nm. These values are shown below, and also the values for IgG molecules and Fab' fragments.

Note: The actual value for the extinction coefficient of Nanogold at 280 nm varies slightly from lot to lot. The most accurate value for the lot you are using will be given in the product specification sheet supplied with your product: use this value for the most accurate labeling calculation.
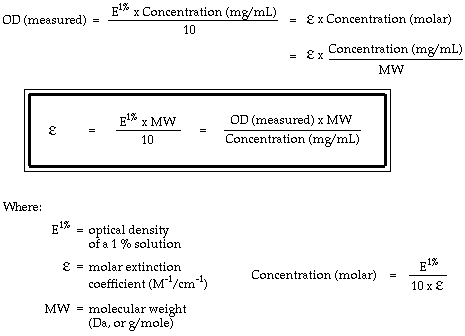
Absorbances of proteins are often given as optical densities (ODs), which expresses absorbance as a funtion of the mass of protein dissolved per unit volume. Usually the literature values (E1%) are for 1 % solutions, (10 mg/mL, or 10 g/L). In order to calculate rates of gold labeling, these must be converted to molar extinction coefficients, epsilon:
Where both the protein and the gold nanoparticle absorb at 280 nm and 420 nm, the observed absorbance at each wavelength is expressed as a pair of simultaneous equations:
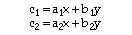
Where:
c1 = measured absorbance at lambda = 280 nm
c2 = measured absorbance at lambda = 420 nm
a1 = extinction coefficient of protein at lambda = 280 nm
a2 = extinction coefficient of protein at lambda = 420 nm
x = concentration of protein (moles/liter)
b1 = extinction coefiicient of gold nanoparticle label at lambda = 280 nm
b2 = extinction coefficient of gold nanoparticle label at lambda = 420 nm
y = concentration of gold nanoparticle label (moles/liter)
Applying Cramer's Rule and solving for x and y in terms of a, b, and c gives:
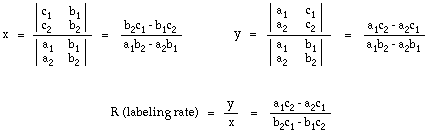
This gives the general solution shown below.
General solution

Labeling of IgG and Fab'
With antibody labeling, the calculation is simplified because these proteins do not absorb at 420 nm; therefore, a2 = 0. This gives the simplified solution below:
Simplified Solution

The following values were measured for UV/visible absorbtion:
280 nm: 0.6524
420 nm: 0.2012
Sample volume: 3.2 mL.
For this sample, the values of the constant terms are:
c1 = 0.6524 (measured absorbance at lambda = 280 nm)
c2 = 0.2012 (measured absorbance at lambda = 420 nm)
a1 = 7.5 X 104 (extinction coefficient of Fab' at lambda = 280 nm)
a2 = 0 (extinction coefficient of Fab' at lambda = 420 nm)
x = concentration of protein (moles/liter)
b1 = 4.12 X 105 (extinction coefiicient of Nanogold® at lambda = 280 nm)
b2 = 1.56 X 105 (extinction coefficient of Nanogold® at lambda = 420 nm)

A hypothetical heme-containing protein with a moleular weight of 65,000 and extinction coefficients of 37,400 at 280 nm and 27,900 at 420 nm is labeled with mono-Sulfo-NHS-undecagold. 2.0 mL of labeled conjugate solution is isolated and absorbances were measured:
280 nm: 0.8705
420 nm: 0.4560
Values for the constants are:
c1 = 0.8705 (measured absorbance at lambda = 280 nm)
c2 = 0.4560 (measured absorbance at lambda = 420 nm)
a1 = 3.74 X 104 (extinction coefficient of protein at lambda = 280 nm)
a2 = 2.79 X 104 (extinction coefficient of protein at lambda = 420 nm)
x = concentration of protein (moles/liter)
b1 = 1.68 X 105 (extinction coefiicient of undecagold at lambda = 280 nm)
b2 = 4.71 X 104 (extinction coefficient of undecagold at lambda = 420 nm)

These results show 1 undecagold nanoparticle for every 5 heme-proteins; this labeling might indicate that the protein forms pentamers, each of which accomodates one gold label - STEM can then be used to investigate further.
|