|
|
Labeling with Gold Nanoparticles:
Introduction: How does gold nanoparticle labeling work?
The advantages of Nanoprobes gold nanoparticles over colloidal gold
![[Undecagold Structure] (3k)](../Images/Au11.gif)
Structure of Monomaleimido Undecagold Gold nanoparticles, or clusters are coordination complexes. They contain a core of gold atoms in a predetermined arrangement; gold atoms at the surface of this core are coordinated by either a tris (aryl) phosphine ligand, or by a halide ion (chloride in the undecagold nanoparticle shown at right) so that the bonding valencies of all the gold atoms are satisfied. As a result, the gold nanoparticle is a stable molecule.
Undecagold and Nanogold®, which is a larger gold nanoparticle containing the same ligands, are uncharged, although other gold nanoparticles such as hexagold (6 gold atoms) and octagold (8 gold atoms) can have an ionic charge.
Problems with Colloidal Gold
In contrast, colloidal gold is not a molecule with a defined structure, but suspended particles with some of the properties of metallic gold. Colloidal gold in solution adopts a negative charge, thought to arise from [AuICl2]- which make up the surface of the particle, as shown below:
![[Colloidal Gold] (7k)](../Images/CollAu.gif)
Colloidal Gold (From: Weiser, H. B.: "Inorganic Colloid Chemistry;" Wiley, New York, NY; Vol. 1, pp. 21-57 (1933).
Because of this charge, colloidal gold has an affinity for many proteins, which tend to be positively charged at neutral or physiological pH. Some proteins, especially antibodies, adsorb strongly to colloidal gold particles: when introduced into a biological specimen which contains binding sites, they carry the colloidal gold particle to this site where it may be visualized. However, this affinity for proteins, or "stickiness," means that the colloidal gold particles can also adsorb to other components of the system, causing false or background labeling. In addition to the conjugated antibody, colloidal gold conjugates must also be stabilized with an inert macromolecule such as bovine serum albumin (BSA), gelatin, carbowax or polyethylene glycol. This increases the size of the probe and can prevent it from penetrating into specimens, for example to label nuclear features. The adsorbed antibody can also dissociate, and free antibodies compete with the conjugate, resulting in less dense labeling. Below are two light micrographs which show a direct comparison between colloidal gold and Nanogold®:
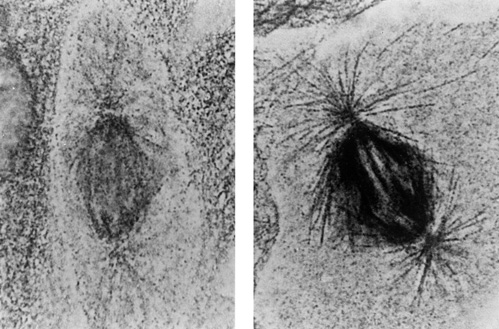
Spindle microtubules labeled with GAM colloidal gold (LEFT) and Fab'-Nanogold® RIGHT)
(Light micrograph courtesey of Dr. D. Vandré and Dr. R. Burry, Ohio State University.
Original magnification = 1300x).
Nanoprobes Gold Nanoparticles: Advantages over Colloidal Gold
Our gold nanoparticles are not adsorbed, but covalently cross-linked to a specific functional group on the targeted biomolecule. For example, monomaleimido undecagold, shown above, reacts only with thiol (sulfhydryl) groups, and therefore attaches to a protein only at a thiol site. This site-specificity allows it to be conjugated at a position remote from the binding site so that it does not interfere with substrate binding; for example, monomaleimido undecagold labels antibody Fab' fragments specifically at the hinge region (which contains the only thiol), is at the other end of the Fab' to the antigen combining region, as shown below:
![[Maleimido Gold Fab' label] (23k)](../Images/Au11SH.gif)
Our gold nanoparticles may be prepared with a number of different reactive groups, for labeling different sites on biological molecules. Undecagold and Nanogold® are also available as mono-amines, which may be used in a variety of cross-linking reactions, or as mono-sulfo-NHS esters, which react with amines.
Site-specific covalent conjugation means that nanoparticles can be used to label a wide variety of biologically active molecules, such as lipids, oligonucleotides, peptides and drugs, which do not adsorb to colloidal gold and hence cannot be colloidal gold-labeled. Reactive forms of Nanogold® and undecagold are supplied lyophilized as labeling reagents, with which our customers can label any molecule with the appropriate chemical group. Once conjugated, they are stable indefinitely, and do not have residual charges which can interact non-specifically with biological systems. Furthermore, the coordinated ligands can be modified to confer desirable properties on the molecule (such as water-solubility).
References
- Immunogold-Silver Staining: Principles, Methods and Applications, M. A. Hayat (Ed.); CRC Press, Boca Raton, FL, 1995. See chapters by: Burry, R. W.; Hainfeld, J. F., and Furuya, F. R. (pp. 71-96); and Hacker, G.
- Hainfeld, J.F. Site specific cluster labels. Ultramicroscopy, 46, 135-144 (1992).
- Hainfeld, J.F. and Furuya, F.R. A 1.4nm Gold cluster covalently attached to antibodies improves immunolabeling, J. Histochem. Cytochem., 40, 177-184 (1992).
- Takizawa, T. and Robinson, J.M. Use of 1.4-nm immunogold particles for immunocytochemistry on ultra-thin cryo- sections. J. Histochem. Cytochem., 42, 1615-1623 (1994).
Further references for labeling reagents.
Next: Improving Labeling Reactions
|