![[Nanoprobes Tech Support (14k)]](../Images/logo_tech.gif)
Updated: March 6, 2008
PRODUCT INFORMATION
EnzMet Western Blot HRP Detection Kit
[EnzMet Western Blot HRP Detection Kit product page]

| Product Name: |
EnzMet Western Blot HRP Detection Kit |
| Catalog Number: |
6002 |
| Revision:: |
1.0 (January 2008) |
Technical Assistance Online
![[6002-PDF]](../Images/pdf.gif) Instructions (PDF) Instructions (PDF)
EnzMet Western Blot HRP Detection Kit is designed for use with horseradish peroxidase (HRP) conjugates for the sensitive and direct visualization of antigens, proteins, and other targets in western and other HRP-based blot applications. EnzMet produces black, sharply defined and non-diffusing stains with high sensitivity and resolution. Staining is permanent, and does not fade with time.
The following materials are sufficient for 150 tissue sections, based upon 200 µL per section:
| EnzMet Western HRP Detect A | 18 mL |
| EnzMet Western HRP Detect B | 6 mL |
| EnzMet Western HRP Detect C | 6 mL |
Refrigerate at 4°C. The product is shipped at ambient temperature.
EnzMet detection utilizes probes labeled with peroxidase enzymes to reduce silver ions to elemental silver, resulting in the deposition of metallic silver particles at sites labeled by peroxidase.1,2 The silver deposits can be directly visualized by eye or by light microscopy for the detection of target proteins,3 DNA4,5 or RNA sequences and other specific binding targets on blots or in biological specimens. EnzMet generates dense, sharply defined, non-diffusing black signals with low background. Its high sensitivity and signal-to-noise ratio enable the detection of targets at low concentration. The non-diffusing and sharply defined stains provide superior spatial resolution for differentiation of targets. The black silver signals are readily distinguishable from other labels and stains, and are not soluble in either aqueous or organic solution.
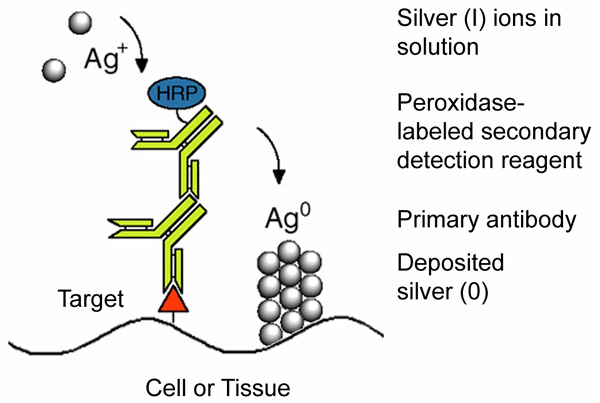
|
Figure 1:
Mechanism of the enzyme metallographic process, showing enzyme-catalyzed deposition of metal from solution at a target site labeled with a primary antibody and peroxidase-labeled secondary antibody.
|
Contents
Note: Volumes indicated below are for one 7 x 8.4 cm2 blot. Volumes can be adjusted for staining one different sized blots or multiple blots.
All EnzMet reagents and other required materials should be equilibrated to room temperature prior to the blotting procedure. All incubations with EnzMet detection should be performed at room temperature with gentle agitation. EnzMet solution B may become slightly yellow-colored during storage. However, the color change will not affect its performance.
For blot staining, follow your standard procedure until incubation with the HRP-conjugated antibody or HRP reagent. After the incubation with the HRP-conjugated antibody or probe, follow these steps:
- Wash with buffer containing 0.1% Tween-20 for 3 x 5 minutes.
Note: Phosphate-buffered saline (PBS), tris buffered saline (TBS) or other wash buffers may be used. Including 0.1 % (w/v) Tween-20 in the wash buffer was found to be helpful in reducing non-specific binding.
- Wash with deionized water for 3 x 5 minutes.
- Shake off excess water. Cover membrane with 6 mL (or 3 volumes) of EnzMet Detect A. Incubate for 4 minutes.
Note: Excess water can lead to the dilution of EnzMet reagents, resulting in weak staining and results which are difficult to reproduce.
- Add 2 mL (or 1 volume) of EnzMet Detect B to the membrane, and gently mix Solutions A and B. Incubate for 4 minutes.
- Add 2 mL (or 1 volume) of EnzMet Detect C to the membrane, and gently mix Solutions A, B and C. Incubate for 9 - 25 minutes, or until satisfactory staining is achieved.
Note: The EnzMet incubation time mainly depends on the target concentrations and staining temperature. Longer incubation may be needed for visualizing low concentration targets. However, longer incubation may lead to some non-specific background staining. The variation of EnzMet staining temperature can affect its silver deposition rate. Lower temperature slows down the deposition process, and thus a longer staining time may be required to reach a certain degree of staining density and sensitivity.
- Wash with deionized water for 3 x 5 minutes.
- Air dry membrane for record.
Contents
EnzMet detection produces black, sharply defined and non-diffusing stains or bands at the sites of antigens or other targets.
For immunoblot HRP detection, we recommend blocking with 5% nonfat dried milk prior to incubation with antibodies. The concentrations and incubation times of primary and secondary antibodies should be tested and adjusted to find the best signal-to-noise ratio. A concentration of 2 µg/mL of HRP-conjugated antibody was found to give satisfactory sensitivity and signal-to-noise ratio. Including 1% nonfat dried milk in incubations with primary and secondary antibodies may be helpful in reducing non-specific background staining.
Contents
- 1. Hainfeld; J. F.; Eisen; R. N.; Tubbs; R. R.; and Powell; R. D.: Enzymatic Metallography: A Simple New Staining Method. Microsc. Microanal.; 8 (Suppl. 2: Proceedings); Lyman; C. E.; Albrecht; R. M.; Carter; C. B.; Dravid; V. P.; Herman; B.; and Schatten; H. (Eds.); Cambridge University Press; New York; NY, 916 CD (2002).
Full paper (HTML)
- Hainfeld, J. F.; Powell, R. D., and Hacker, G. W.: Nanoparticle Molecular Labels. In: Nanobiotechnology, Mirkin, C. A., and Niemeyer, C. M. (Eds): Wiley-VCH, Weinheim, Germany; ch. 23, pp. 353-386 (2004).
- Tubbs, R.; Pettay, J.; Powell, R.; Hicks, D. G.; Roche, P.; Powell, W.; Grogan, T, and Hainfeld, J. F.: High-Resolution Immunophenotyping of Subcellular Compartments in Tissue Microarrays by Enzyme Metallography. Appl. Immunohistochem. Mol. Morphol., 13, 371-375 (2005).
- Tubbs, R.; Pettay, J.; Hicks, D.; Skacel, M.; Powell, R.; Grogan, T., and Hainfeld, J.: Novel bright field molecular morphology methods for detection of HER2 gene amplification. J. Mol. Histol., 35, 589-594 (2004).
- Powell, R. D.; Pettay, J. D.; Powell, W. C.; Roche, P. C.; Grogan, T. M.; Hainfeld, J. F., and Tubbs, R. R.: Metallographic in situ hybridization. Hum. Pathol., 38, 1145-1159 (2007).
Technical Assistance Available.
For a complete list of references citing this product, please visit our References page on our web site.
Contents
|