![[Nanoprobes Tech Support (14k)]](../Images/logo_tech.gif)
Updated: March 6, 2008
PRODUCT INFORMATION
EnzMet HRP Detection Kit for IHC / ISH
[EnzMet HRP Detection Kit for IHC / ISH product page]

| Product Name: |
EnzMet HRP Detection Kit for IHC / ISH |
| Catalog Number: |
6001 |
| Revision:: |
1.1 (January 2008) |
Technical Assistance Online
![[6001-PDF]](../Images/pdf.gif) Instructions (PDF) Instructions (PDF)
EnzMet IHC/ISH HRP Detection Kit is designed for use with horseradish peroxidase (HRP) conjugates for the sensitive, high-resolution visualization of target proteins and nucleic acids in biological specimens. EnzMet produces black, sharply defined and non-diffusing stains with high sensitivity and resolution. Staining is permanent, does not fade with time, and is readily distinguished from commonly used stains and counterstains.
The kit is intended for research use only. It is not recommended, approved or intended for in vitro diagnosis of disease in humans or animals. Do not use internally or externally in humans or animals. Not to be used for in vivo therapeutic or human uses, or any unauthorized commercial uses. This EnzMet kit is specially optimized for standard, manual immunohistochemical (IHC) and in situ hybridization (ISH) detection.
The following materials are sufficient for 150 tissue sections, based upon 200 µL per section:
| EnzMet IHC/ISH HRP Detect A | 18 mL |
| EnzMet IHC/ISH HRP Detect B | 6 mL |
| EnzMet IHC/ISH HRP Detect C | 6 mL |
Refrigerate at 4°C. The product is shipped at ambient temperature.
EnzMet detection utilizes peroxidase enzymes to reduce silver ions to elemental silver, resulting in the deposition or accumulation of silver metal particles at sites labeled by peroxidase.1,2 The silver deposits can be directly visualized by light microscopy for the detection of target proteins,3 DNA4,5 or RNA sequences and other specific binding targets in paraffin-embedded tissues, cryostat sections and cell preparations. EnzMet generates dense, sharply defined, non-diffusing black signals with low background. Its high sensitivity and signal-to-noise ratio enable the detection of targets at low concentration and the evaluation of samples at low magnification. The non-diffusing and sharply defined stains provide superior spatial resolution for differentiation of cellular compartments, precise localization and enumeration of targets. The black silver signals are readily distinguished from other stains,6 and are compatible with both organic and aqueous based counterstains, dehydration protocols, and various mounting media.
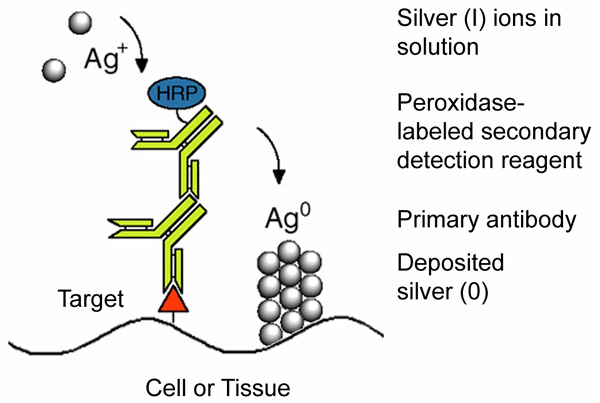
|
Figure 1:
Mechanism of the enzyme metallographic process, showing enzyme-catalyzed deposition of metal from solution at a target site labeled with a primary antibody and peroxidase-labeled secondary antibody.
|
Contents
Note: Volumes indicated below are based upon 200 µL per one slide. Volumes can be adjusted for fully covering your staining area.
All EnzMet reagents should be equilibrated to room temperature prior to the staining procedure. All incubation steps in EnzMet detection should be performed at room temperature. EnzMet solution B may become slightly yellow-colored during storage. However, the color change will not affect its performance.
For IHC and ISH staining, follow your standard procedure until incubation with the HRP-conjugated antibody or HRP reagent. After incubation with the HRP-conjugated antibody or probe, follow these steps:
- Wash the specimen with buffer for 2 x 5 minutes. Including 0.1 % (w/v) Tween-20 in the wash buffer has been found to be helpful in reducing non-specific binding.
- Wash the specimen with deionized water for 3 x 5 minutes.
- Tap off excess water. Cover specimen with 120 µL (or 3 volumes) of EnzMet Detect A. Incubate for 2 minutes.
Note: Excess water can lead to the dilution of EnzMet reagents, resulting in weak staining and results which are difficult to
reproduce.
- Add 40 µL (or 1 volume) of EnzMet Detect B to the specimen, and agitate gently to mix Solutions A and B. Incubate for 2
minutes.
- Add 40 µL (or 1 volume) of EnzMet Detect C to the specimen, and agitate gently to mix Solutions A, B and C. Incubate for 8 - 15 minutes, or until satisfactory staining is achieved.
Note: The EnzMet incubation time depends on the target concentrations and staining temperature. Longer incubation may be needed for visualizing low abundance targets. However, longer incubation may lead to some non-specific background staining. Variation in the temperature at which EnzMet staining is done can affect its silver deposition rate. Lower temperatures slow down the deposition process, and thus longer staining times may be required.
- Wash with deionized water for 3 x 5 minutes.
- Counterstain and dehydrate. The EnzMet stain is compatible with both aqueous- or organic-based counterstains.
- Coverslip with aqueous- or organic-based mounting media.
Contents
In IHC and ISH procedures, the EnzMet detection produces black, sharply defined and non-diffusing stains at the sites of the target antigens or nucleic acids.
For IHC applications, the endogenous peroxidase or pseudoperoxidase activity can be inhibited by incubating specimens with 3% hydrogen peroxide solution for ten minutes at room temperature before application of the primary antibody. The concentrations and incubation time for primary and secondary antibodies should be tested and adjusted to find the best signal-to-noise ratio.
The hybridization, denaturation and stringency conditions of ISH procedures vary according to the probe/target combination and the specimen used. Users should refer to the instructions provided with their probe for specific information regarding hybridization times and temperatures, stringent wash temperatures, and denaturation condition for double-stranded DNA probes.
Sample protocols for IHC and ISH are given below. These protocols are intended as examples rather than specifications or directions. When constructing a protocol for the use of this reagent, we recommend that the steps before the EnzMet application be conducted as directed by the supplier of each component if these differ significantly from the directions below.
EnzMet Detection of epithelial Cytokeratins in human bladder tumor tissues
Note: Volumes indicated below are based upon 200 µL per one section.
Materials Required
- Paraffin embedded human bladder tumor tissue
- Xylene
- Ethanol, 95%, 90%, 85%, and 70% ethanol
- 3% hydrogen peroxide solution
- TBST: 20 mM Tris pH7.6, 150 mM NaCl and 0.1 % Tween-20
- 0.1% protease solution, pH 7.8
- Monoclonal mouse anti-human cytokeratin clone: AE1/AE3 antibody (Dako, Ref. # M3515).
- Bovine serum albumin
- Peroxidase (HRP) conjugated goat anti-mouse IgG (Jackson Immunochemicals, Cat. # 71556).
- Nuclear Fast Red Stain (NewComer Supply, Cat. # 1255)
- Cytoseal-60 (Richard Allan Scientific, Cat. # M7630-1A)
Procedure
The paraffin embedded tissue slide and all materials are equilibrated to room temperature prior to the IHC procedure.
- Deparaffinize by sequentially dipping slide in xylene (2 x 10minutes), absolute ethanol (2 x 3 minutes), and 95%, 90%, 85%, and 70% ethanol (3 minutes each), respectively.
- Rehydrate by incubating in water for 3 minutes and then in TBST for 5 minutes.
- Incubate with 200µL of 3% H2O2 solution in TBST for 10 min at room temperature (RT) in a moisture chamber.
- Incubate the slide with TBST for 5 minutes at RT.
- 5. Incubate with 200 ?L of 0.1% protease solution (pH 7.8) for 5 minutes at 37C in a moisture chamber.
- Incubate with TBST for 2 x 5 minutes at RT.
- Incubate with 200 ?L of monoclonal mouse anti-human AE1/AE3 antibody solution (1:1500 in TBST containing 0.3% BSA) for 30 minutes at 37°C in a moisture chamber.
- Incubate with TBST for 2 x 5 minutes at RT.
- Incubate with 200µL of HRP-conjugated goat anti mouse IgG solution (1:1500 in TBST containing 0.3% BSA) for 120 minutes at 37°C in a moisture chamber.
- Incubate with TBST for 2 x 5 minutes at RT.
- Perform EnzMet staining procedure according to the procedure given above.
- After 10 minutes of EnzMet incubation, wash with deionized water for 3 x 5 minutes.
- Counterstain with Nuclear Fast Red for 8 minutes.
- Optional: dehydrate the tissue section by reversing the deparaffinization and hydration steps.
- Coverslip with Cytoseal-60.
Reagents and Equipment
- Phosphate buffered saline (PBS): 20 mM sodium phosphate buffer, with 150 mM NaCl, adjusted to pH 7.4.
- Proteinase K.
- In situ hybridization probe labeled with a suitable detection label (biotin, fluorescein, et.).
- Coplin jar or incubation chamber.
- Coverslips.
- Standard Sodium Citrate (SSC) buffer: 300 mM NaCl, 30 mM sodium citrate, pH 7.0
- Washing buffer - PBST: PBS containing 0.1% Tween-20.
- Primary antibody or probe (peptide or protein).
- Peroxidase conjugated secondary antibody.
- EnzMet Solutions A, B and C.
- Deionized water for rinsing.
Procedure
Starting with paraffin-embedded section, and using a biotinylated hybridization probe:
- Deparaffinize in xylene, 3 x 7 minutes.
- Redehydrate with 100% alcohol, 2 x 1 min, followed by 95% alcohol, 2 x 1 minute, then water soak, 5 minutes.
- Apply target retrieval solution for 40 minutes at 95°C or as directed by the manufacturer; then cool down to room temperature for 20 minutes.
- Shake off the excess and apply Proteinase K dissolved to 0.05% in PBS and incubate at 37°C for about 6 to 8 minutes. The duration will vary depending upon the target retrieval conditions and time, and should be tested very carefully, depending on tissue, fixation and other factors.
- To inactivate any endogenous peroxidase, apply 3% H2O2, 20 minutes at room temperature.
- Rinse in deionized water.
- Dehydrate through 80% alcohol (1 minute), 95% alcohol (1 minute), then 100% alcohol (1 minute). Allow to air dry.
- Add 10 µL of hybridization probe; use normal dilution or as directed by manufacturer. Coverslip and seal for hybridization.
- Codenature 6 minutes at 90°C.
- Hybridize with probe mixture at 37°C for at least 4 hours or overnight, depending on the protocol for your probes.
- Remove coverslip: soak in 2x Standard Sodium Citrate (SSC) buffer for 5 minutes.
- Stringent Wash: SSC, 72°C for 5 minutes.
- Rinse in deionized water, 3 x 2.5 minutes.
- Wash in PBS, 3 X 2.5 minutes.
- Apply non-specific blocking Solution (10 minutes).
- Apply primary antibody. We suggest using a dilution 5-fold greater than that used or directed when the final detection is with DAB or similar organic chromogens: for example, if DAB detection protocol recommends 100-fold dilution of the primary antibody, use a 500-fold dilution, then adjust up or down as necessary. Incubate for 1 hour.
- Wash with PBS containing 0.1% Tween 20, 3 x 5 minutes.
- Apply horseradish peroxidase-labeled secondary antibody. Use the same dilution and incubation time as you would for detection using organic chromogens such as DAB.
- Wash with PBS containing 0.1% Tween 20, 3 x 5 minutes.
- Rinse in deionized water, 3 x 5 minutes.
- Develop with EnzMet metallographic substrate as directed in separate protocol.
Contents
- 1. Hainfeld; J. F.; Eisen; R. N.; Tubbs; R. R.; and Powell; R. D.: Enzymatic Metallography: A Simple New Staining Method. Microsc. Microanal.; 8 (Suppl. 2: Proceedings); Lyman; C. E.; Albrecht; R. M.; Carter; C. B.; Dravid; V. P.; Herman; B.; and Schatten; H. (Eds.); Cambridge University Press; New York; NY, 916 CD (2002).
Full paper (HTML)
- Hainfeld, J. F.; Powell, R. D., and Hacker, G. W.: Nanoparticle Molecular Labels. In: Nanobiotechnology, Mirkin, C. A., and Niemeyer, C. M. (Eds): Wiley-VCH, Weinheim, Germany; ch. 23, pp. 353-386 (2004).
- Tubbs, R.; Pettay, J.; Powell, R.; Hicks, D. G.; Roche, P.; Powell, W.; Grogan, T, and Hainfeld, J. F.: High-Resolution Immunophenotyping of Subcellular Compartments in Tissue Microarrays by Enzyme Metallography. Appl. Immunohistochem. Mol. Morphol., 13, 371-375 (2005).
- Tubbs, R.; Pettay, J.; Hicks, D.; Skacel, M.; Powell, R.; Grogan, T., and Hainfeld, J.: Novel bright field molecular morphology methods for detection of HER2 gene amplification. J. Mol. Histol., 35, 589-594 (2004).
- Powell, R. D.; Pettay, J. D.; Powell, W. C.; Roche, P. C.; Grogan, T. M.; Hainfeld, J. F., and Tubbs, R. R.: Metallographic in situ hybridization. Hum. Pathol., 38, 1145-1159 (2007).
- Downs-Kelly, E.; Pettay, J.; Hicks, D.; Skacel, M.; Yoder, B.; Rybicki, L.; Myles, J.; Sreenan, J.; Roche, P.; Powell, R.; Hainfeld, J.; Grogan, T., and Tubbs, R.: Analytical Validation and Interobserver Reproducibility of EnzMet GenePro: A Second-Generation Bright-Field Metallography Assay for Concomitant Detection of HER2 Gene Status and Protein Expression in Invasive Carcinoma of the Breast. Am. J. Surg. Pathol., 29, 1505-1511 (2005).
Technical Assistance Available.
For a complete list of references citing this product, please visit our References page on our web site.
Contents
|