Prepare the smallest gold immunoprobe with your own primary antibody
for unsurpassed resolution, penetration and localization, or label a variety of other biological molecules.
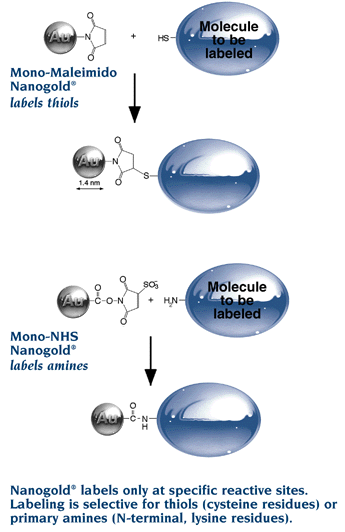
- Our precision 1.4 nm Nanogold® label, with a single reactive maleimide
- Reacts with thiols (sulfhydryls) under mild conditions (pH 6.0 to 7.0).
- Covalently labels Fab’, IgG, proteins or peptides containing cysteine, and other molecules with sulfhydryls
- Use our unique gold labeling reagents just as you would use fluorescent labeling reagents
Easy to use:
Simply mix reconstituted Monomaleimido Nanogold® with the target molecule for one hour at room temperature or 16 h in the refrigerator, then chromatographically purify the product by gel filtration, ion exchange chromatography or hydrophobic interaction chromatography. The maleimide on the 1.4 nm Nanogold® particle specifically reacts with a free sulfhydryl under mild conditions to produce a stable covalent bond.
Applications:
- Create your own small gold probes by linking Nanogold® to your primary antibody Fab’ fragments. [References]
- Label other proteins directly for high resolution site specific identification.
- Label peptides containing cysteine (see Gregori, L., et al.; J. Biol. Chem., 272, 258-262 (1997)). [References]
- Label 5′-thiolated oligonucleotides directly with Nanogold® (see Alivisatos, A. P., et al.; Nature, 382, 609 (1996)). [References]
Monomaleimido Nanogold® labeling of the phosphorylase kinase complex
![STEM images of phosphorylase kinase complex with Nanogold®-labeled d subunit. Phosphorylase-b kinase was incubated with excess exogenous calmodulin and a threshold concentration of urea to promote exchange of calmodulin for the d subunit and size exclusion-HPLC was used to remove the excess calmodulin from the enzyme. Using metabolically labeled [35S]calmodulin to allow quantification, an exchange of approximately 10% of all d subunits within 1 h was measured. The exchanged enzyme retained full catalytic activity. Calmodulins that were then derivatized with Monomaleimido-Nanogold® and exchanged under the conditions described above. Analysis of STEM images of these Nanogold®-exchanged images allowed the placement of the d subunit within the phosphorylase kinase holoenzyme. From: Traxler KW, Norcum MT, Hainfeld JF, Carlson GM. Direct Visualization of the Calmodulin Subunit of Phosphorylase Kinase via Electron Microscopy Following Subunit Exchange. J Struct Biol. 2001 Sep;135(3):231-8.](https://nanoprobes.com/wp-content/uploads/monomaleimido-nanogold-labeled-phosphorylase-kinase-STEM.jpg)
STEM images of phosphorylase kinase complex with Nanogold®-labeled d subunit.
Phosphorylase-b kinase was incubated with excess exogenous calmodulin and a threshold concentration of urea to promote exchange of calmodulin for the d subunit and size exclusion-HPLC was used to remove the excess calmodulin from the enzyme. Using metabolically labeled [35S]calmodulin to allow quantification, an exchange of approximately 10% of all d subunits within 1 h was measured. The exchanged enzyme retained full catalytic activity. Calmodulins that were then derivatized with Monomaleimido-Nanogold® and exchanged under the conditions described above. Analysis of STEM images of these Nanogold®-exchanged images allowed the placement of the d subunit within the phosphorylase kinase holoenzyme.
From: Traxler KW, Norcum MT, Hainfeld JF, Carlson GM. Direct Visualization of the Calmodulin Subunit of Phosphorylase Kinase via Electron Microscopy Following Subunit Exchange. J Struct Biol. 2001 Sep;135(3):231-8.
1.4 nm Nanogold® Labeling Reagents
Nanogold® is a better gold label.
The 1.4 nm Nanogold® particle is a gold compound: it is not just adsorbed to proteins, like colloidal gold, but covalently reacts at specific sites under mild buffer conditions. This gives a well defined product that can be purified chromatographically.
Nanogold® brings the versatility of fluorescent conjugation to gold labeling.
Label virtually any molecule
Label any molecule with a suitable reactive group: oligonucleotides, lipids, peptides, proteins, enzyme inhibitors and others. This is a big improvement over colloidal gold, which may be adsorbed only to antibodies and a limited range of proteins and peptides.
Nanogold® is small and highly uniform in size, in sharp contrast to small colloidal gold preparations (most commonly used “1 nm” colloidal golds actually range from 1 to 3 nm).
Label your own biomolecules with our Nanogold® labeling reagents,
or check out our range of antibody IgG, Fab’ and streptavidin Conjugates with Nanogold®.

Left: Silver-enhanced Nanogold®-labeled K+ channel Kv2.1 subunit in rat brain, X15,000 (J.-H. Tao-Cheng, NIH).
Right: Darkfield STEM micrographs of Maleimido Nanogold® labeled A β peptide targeting proteasomes; sample was stained with methylamine vanadate. White dots are 1.4 nm Nanogold®.From Gregori, L., Hainfeld, J.F., Simon, M.N., and Goldgaber, D. (1997). Binding of amyloid beta protein to the 20S proteasome. J. Biol. Chem. 272, 58-62.
Easily enhanced for electron microscopy, light microscopy, cryo-EM, blots…
Try our precision nanoparticle developers for slow development with low background!
Features and Advantages
- Unparalleled penetration of conjugates -up to 40 µm!
- Higher density of immunolabeling than with larger gold probes.
- Can be conjugated to any molecule with a suitable reactive group. Available with different reactivities.
- Extremely uniform 1.4 nm gold particle.
- Label at specific sites which do not obstruct native reactivity.
- Close to stoichiometric labeling.
- Reacts under mild, neutral conditions.
- Conjugates are easily isolated by gel filtration.
- Conjugates are stable to a wide range of pH and ionic strengths.
- High stability: conjugates show unchanged reactivity after storage for a year.